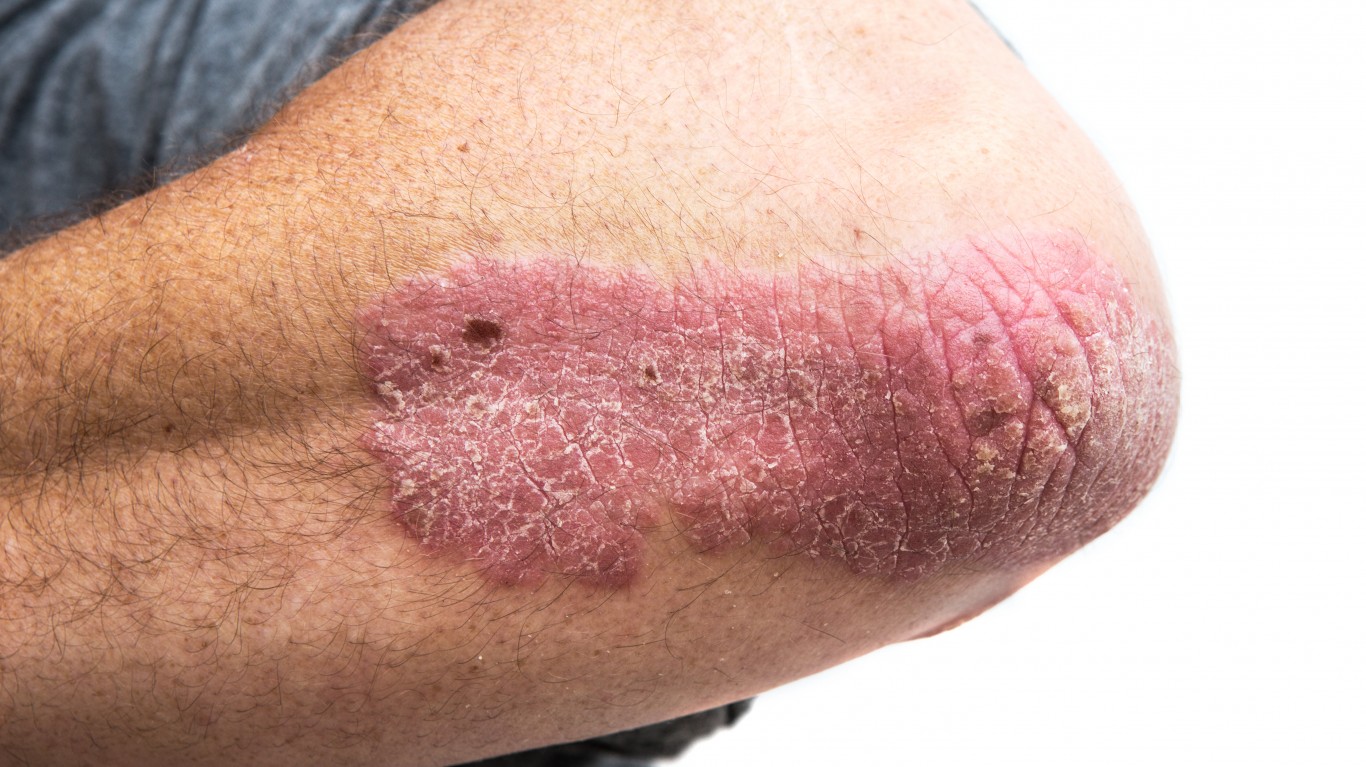
Vitae Pharmaceuticals Inc. (NASDAQ: VTAE) watched its shares absolutely take off on Thursday morning following the release of positive results from its mid-stage clinical trial. The company announced positive top-line results from its Phase 2a proof-of-concept clinical trial of VTP-43742 in psoriatic patients.
Prior to the jump in shares, Vitae’s stock was actually down year to date about 80% and trading at a 52-week low.
For some background: VTP-43742 is Vitae’s wholly owned, first-in-class, orally active RORγt inhibitor with the potential to transform the treatment of multiple autoimmune disorders, including psoriasis, through the potent inhibition of IL-17 secretion from Th17 cells and blocking the action of IL-23.
VTP-43742 demonstrated a clear signal of efficacy, with patients in the 350 mg dose group achieving a 24% reduction in the Psoriasis Area Severity Index (PASI) score relative to placebo. In the 700 mg dose group, patients achieved a 30% placebo-adjusted PASI score reduction. For both doses, the company observed clinically relevant and statistically significant reductions relative to baseline values.
[nativounit]
Jeff Hatfield, president and CEO of Vitae, commented:
We believe these data validate RORγt as an exciting and novel therapeutic target for the treatment of psoriasis and other autoimmune disorders. While the autoimmune market is currently dominated by injectable antibody therapy, we believe VTP-43742 has the potential to expand utilization of oral therapy in a variety of autoimmune disorders, such as psoriasis, psoriatic arthritis, rheumatoid arthritis, multiple sclerosis and inflammatory bowel disease with an effective, safe and well tolerated, once-a-day agent.
Dr. Richard Gregg, chief scientific officer of Vitae, added:
We plan to advance VTP-43742 into a larger scale 16 week trial in the second half of 2016 to continue to assess the efficacy, safety and tolerability of our first-in-class drug candidate.
Shares of Vitae closed Wednesday down 7.6% at $4.11, with a consensus analyst price target of $21.67 and a 52-week trading range of $4.08 to $18.71. Following the release of the results, the stock was up 80% at $7.40 in early trading indications Thursday.