Moderna Inc (NASDAQ:MRNA | MRNA Price Prediction) has delivered a 37% gain year-to-date, dramatically outpacing the biotech sector’s 0.4% YTD return. In fact, Moderna has the 12th-best gains in the entire S&P 500 so far in 2026!
The rally reflects strategic wins and pipeline progress, though recent regulatory turbulence has tested investor conviction. Let’s dive into why Moderna is one of the market’s biggest winners so far this year.
Earnings Beat Fuels Momentum
Moderna’s fourth quarter 2025 results, filed via 8-K in January 2026, showed the company executing on its cost-cutting roadmap. CEO Stéphane Bancel emphasized the operational shift: “strengthened commercial execution, successfully launched third product, reduced annual operating expenses by approximately $2 billion.” That expense reduction is an important metric for investors to track because cost-cutting was necessary. The company burned through $4.5 billion in R&D during 2024 while revenue collapsed 53% year-over-year to $3.2 billion.
Third quarter 2025 provided the blueprint. Revenue hit $1.02 billion, beating the Street’s $904 million estimate, while the company posted a loss of $0.51 per share versus expectations of a $2.15 loss. The stock jumped 43% from $24.03 in early November to $34.33 by mid-January.
Mexico Deal Expands Geographic Footprint
On February 10, 2026, Moderna announced a five-year agreement with the Mexican government, BIRMEX, and Laboratorios Liomont. The deal includes technology transfer for domestic COVID-19 vaccine production and supply commitments for Moderna’s respiratory vaccine portfolio. This represents a revenue bridge beyond the U.S. market, where COVID vaccine demand has normalized. The partnership supports Mexico’s “Plan Mexico” initiative for local mRNA manufacturing infrastructure.
Pipeline Monetization Gains Traction
The February 6 Recordati collaboration delivered $50 million upfront for mRNA-3927, targeting propionic acidemia. With up to $110 million in near-term milestones plus royalties, the structure demonstrates Moderna can extract value from rare disease assets without bearing full commercialization costs. This matters because the company is targeting cash breakeven by 2028, and partnership economics accelerate that timeline.
FDA Setback Tests Conviction
The rally hit turbulence on February 10th when the FDA issued a refusal-to-file letter for mRNA-1010, Moderna’s seasonal influenza vaccine. The rejection centered on study design, specifically the choice of comparator vaccine in Phase 3 trials, not safety or efficacy concerns. The stock dropped 8% on the news, though management emphasized the application remains under review in the EU, Canada, and Australia. Moderna stated this does not impact 2026 financial guidance, suggesting the flu vaccine was not embedded in near-term revenue projections.
What’s Next for 2026
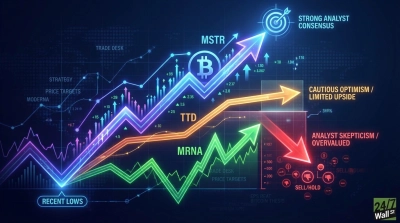
Management is guiding for approximately 10% revenue growth in 2026, with potential first approvals for flu and flu/COVID combination products. The RSV vaccine mRESVIA has Phase 3 data expected in 2026. Wall Street’s analyst target of $38.80 sits just below the current price of $40.51, with 18 of 24 analysts rating the stock a Hold.
The company’s $425 million in annual interest income provides a cash cushion, but the path to profitability hinges on expanding beyond COVID vaccines. The Mexico deal and Recordati partnership show progress. The FDA setback is a reminder that regulatory performance remains the gating factor for this rally to extend beyond the first quarter.